By Michael miskimmon
Atomic Structure
Name : titanium
symbol: Ti (capital t lower case I)
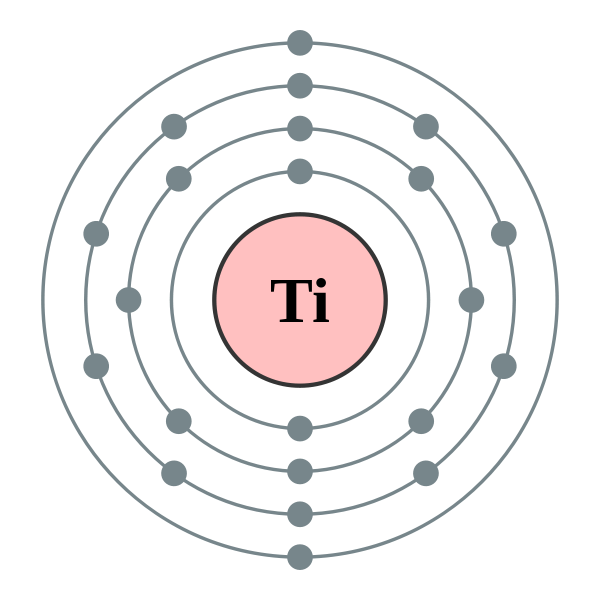
Atomic number: 22
Number of Protons: 22
number of Neutrons: 26
Number of electrons: 22
atomic mass: 47.867 amu
Standard state: solid at 298.0 kelvin
Density: 4.506 g/cm^-3
Melting Point: 1941.0 kelvin, 1660.0 degrees Celsius
Boiling Point: 3560.o kelvin, 3287.0 degrees Celsius
Cost per ton: 10,845.00 us dollars
Period in Periodic table: 4
Family: Transition Metals
color: Metallic White
classification: Metal
discoverer: William gregor (1791)
Named by: Martin Heinrich Klaproth
Origin of Name: For the titans of greek mythology
symbol: Ti (capital t lower case I)
Atomic number: 22
Number of Protons: 22
number of Neutrons: 26
Number of electrons: 22
atomic mass: 47.867 amu
Standard state: solid at 298.0 kelvin
Density: 4.506 g/cm^-3
Melting Point: 1941.0 kelvin, 1660.0 degrees Celsius
Boiling Point: 3560.o kelvin, 3287.0 degrees Celsius
Cost per ton: 10,845.00 us dollars
Period in Periodic table: 4
Family: Transition Metals
color: Metallic White
classification: Metal
discoverer: William gregor (1791)
Named by: Martin Heinrich Klaproth
Origin of Name: For the titans of greek mythology
Physical Properties
- Titanium is recognized for its high strength-to-weight ratio.
- It is a strong metal with low density, is ductile, lustrous, and is metallic white in color.
- Titanium has fairly low electrical and thermal conductivity.
- Titanium is fairly hard and non-magnetic.
- Titanium structures also have a fatigue limit which guarantees longevity in most applications.
- Titanium is 60% more dense than aluminum, but more than twice as strong as its strongest alloy, 6061-t6 aluminum alloy.
- Titanium is as strong as some steels, and is 45% lighter.
chemical properties
- Titanium oxidizes immediately upon exposure to air
- Titanium readily reacts with oxygen, forming Titanium dioxide, which is also the most commonly used compound of titanium (picture on top right)
- Titanium exhibits excellent resistance to corrosion in sea water, aqua regia, and chlorine.
- Titanium is thermodynamically a very reactive metal For example, Titanium burns before its melting point is reached. Because of this, melting is only possible in an inert atmosphere or in a vacuum
- Titanium is one of the few elements that burns in pure nitrogen gas
why should I buy titanium?
pigments, additives, and coatings - 95% of titanium ore extracted from the earth's crust is destined for refinement into titanium dioxide which is a permanent pigment used in paper, paints (mainly for marine use), toothpaste, and plastics. It is also used in cement and gemstones. Titanium is used as a strengthening agent in composite fishing rods and golf clubs. Titanium is also used as a pigment in sunscreen.
Aerospace and Marine - Due to titanium's high strength to density ratio, high corrosion ratio, fatigue resistance, high crack resistance and ability to withstand high temperatures, titanium alloys are used in aircraft, armor plating, naval ships, spacecraft, and missiles. Titanium is used in aircraft engines and frames. Titanium alloys were used in creating the sr-71 "blackbird, and now titanium alloys are found in most commercial aircraft. Titanium is also used to make propeller shafts and divers' knives. The soviet union was even able to make submarines with hulls made of titanium.
Consumer - Titanium is used in automotive applications, especially in automobile and motorcycle racing. For example Pagani and Ariel use titanium in the bodies of their cars. Titanium is used in many sporting goods, such as, tennis rackets, golf clubs, lacrosse sticks, football helmets, and bicycle frames. Many backpackers also use titanium equipment , including lanterns and cookware.
Jewelry - Because of its durability has become very popular for designer jewelry(especially rings). Titanium is also alloyed with gold to produce an alloy that can be marketed as 24 carat gold, as the 1% of alloyed Ti is insufficient to require a lesser mark. The resulting alloy is roughly the hardness of 14-carat gold and thus is more durable than a pure 24-carat gold item would be.
Medical - Because Titanium is Biocompatible (it is non=toxic and is not rejected by the body), is used in surgical implements and implants, including hip balls and sockets that can stay in place for 20 years. Titanium also has the ability to osseointegrate (functional connection between living bone and artificial implants), which enables the use of dental implants that can remain in place for 30 years.
Nuclear waste storage - Due Titanium's extreme corrosion resistance, titanium containers are being used for the long term storage of nuclear waste; these containers last close to 100,000 years.
Aerospace and Marine - Due to titanium's high strength to density ratio, high corrosion ratio, fatigue resistance, high crack resistance and ability to withstand high temperatures, titanium alloys are used in aircraft, armor plating, naval ships, spacecraft, and missiles. Titanium is used in aircraft engines and frames. Titanium alloys were used in creating the sr-71 "blackbird, and now titanium alloys are found in most commercial aircraft. Titanium is also used to make propeller shafts and divers' knives. The soviet union was even able to make submarines with hulls made of titanium.
Consumer - Titanium is used in automotive applications, especially in automobile and motorcycle racing. For example Pagani and Ariel use titanium in the bodies of their cars. Titanium is used in many sporting goods, such as, tennis rackets, golf clubs, lacrosse sticks, football helmets, and bicycle frames. Many backpackers also use titanium equipment , including lanterns and cookware.
Jewelry - Because of its durability has become very popular for designer jewelry(especially rings). Titanium is also alloyed with gold to produce an alloy that can be marketed as 24 carat gold, as the 1% of alloyed Ti is insufficient to require a lesser mark. The resulting alloy is roughly the hardness of 14-carat gold and thus is more durable than a pure 24-carat gold item would be.
Medical - Because Titanium is Biocompatible (it is non=toxic and is not rejected by the body), is used in surgical implements and implants, including hip balls and sockets that can stay in place for 20 years. Titanium also has the ability to osseointegrate (functional connection between living bone and artificial implants), which enables the use of dental implants that can remain in place for 30 years.
Nuclear waste storage - Due Titanium's extreme corrosion resistance, titanium containers are being used for the long term storage of nuclear waste; these containers last close to 100,000 years.
Examples of Cool things made with titanium
References
Bentor, Yinon. Chemical Element.com - Titanium. Oct.6, 2013
http://www.chemicalelements.com/elements/ti.html.
"Titanium." WebElements Periodic Table of the Elements. N.p., n.d. Web.
07 Oct. 2013.
Donachie, Matthew J., Jr. (1988). TITANIUM: A Technical Guide. Metals Park, OH: ASM
International. p. 11
http://www.chemicalelements.com/elements/ti.html.
"Titanium." WebElements Periodic Table of the Elements. N.p., n.d. Web.
07 Oct. 2013.
Donachie, Matthew J., Jr. (1988). TITANIUM: A Technical Guide. Metals Park, OH: ASM
International. p. 11